Nursing diagnoses related to Chlamydia infection reflect not only the physiological effects of the disease but also its psychological, emotional, and social consequences. Ineffective healing is a primary concern, particularly in cases where infection persists because of delayed diagnosis, untreated exposure, or poor adherence to therapy. The intracellular nature of Chlamydia trachomatis contributes to sustained inflammation and tissue injury, especially within the reproductive tract, which may compromise normal healing and increase the risk of long-term complications (Murray & McKay, 2021; O’Connell & Ferone, 2016; Hocking et al., 2023). Nurses play an essential role in monitoring clinical progress, reinforcing adherence to prescribed antimicrobial regimens, and identifying early signs of complications that may delay recovery (Workowski et al., 2021; Hao et al., 2025).
Deficient knowledge is also commonly observed among patients diagnosed with Chlamydia, particularly regarding transmission routes, the asymptomatic nature of infection, and the importance of partner treatment. Many patients remain unaware of the possible long-term consequences of untreated infection, including infertility, chronic pelvic pain, and recurrent reproductive morbidity (Hocking et al., 2023; Pelvic inflammatory disease, 2012; Zhao et al., 2025). This gap in understanding can significantly influence health-seeking behavior, treatment adherence, and follow-up participation. Nursing interventions therefore place strong emphasis on patient education, using clear, culturally sensitive, and nonjudgmental communication to improve awareness, encourage preventive practices, and reduce the risk of reinfection (Workowski et al., 2021; Hao et al., 2025).
Anxiety frequently accompanies the diagnosis of a sexually transmitted infection. Patients may experience fear related to stigma, disclosure to sexual partners, reproductive consequences, or the impact of diagnosis on future relationships. Emotional distress may also arise from uncertainty surrounding prognosis or concerns regarding reinfection and social judgment (Crichton et al., 2015; Zhao et al., 2025). Nurses must assess these psychological responses and provide supportive care through reassurance, counseling, and, when appropriate, referral for further psychosocial support. Addressing anxiety is important not only for emotional well-being but also for improving patient engagement and treatment compliance.
Another relevant nursing diagnosis is ineffective body defense, as Chlamydia infection may alter local mucosal immunity and increase vulnerability to secondary infection or persistent inflammation. Disruption of epithelial barriers and immune activation may weaken the body’s protective mechanisms, particularly in the genital tract (Redgrove & McLaughlin, 2014; Murray & McKay, 2021). Nursing care in such cases includes monitoring for signs of co-infection, promoting immune-supportive practices, and facilitating timely medical intervention when complications arise.
Sexual dysfunction may also occur as a consequence of physical discomfort, emotional stress, fear of infecting a partner, or stigma associated with sexually transmitted infections. Some patients may begin to avoid intimacy because of pain, embarrassment, or concerns regarding disclosure and transmission, which can negatively affect both relationships and quality of life (Hocking et al., 2023; Workowski et al., 2021). Nurses should create a safe and respectful environment in which patients feel comfortable discussing sexual health concerns, while also offering guidance on safe sexual practices and emotional support aimed at restoring confidence and well-being.
Pain is another important nursing concern, particularly in patients with pelvic inflammatory disease, urethral involvement, or associated inflammatory complications. Pain may range from mild discomfort to severe pelvic, abdominal, or genitourinary pain, with meaningful effects on daily functioning and emotional well-being (Pelvic inflammatory disease, 2012; Ekabe et al., 2017). Effective nursing care requires accurate pain assessment, timely medication administration, ongoing evaluation of treatment response, and education regarding warning symptoms that may indicate worsening disease.
In addition, low self-esteem may emerge due to the stigma commonly associated with sexually transmitted infections. Patients may experience shame, guilt, embarrassment, or self-blame, all of which may influence mental health and interpersonal functioning (Crichton et al., 2015). Nursing care should therefore include emotional support, therapeutic communication, and reassurance that Chlamydia is both common and treatable, and that seeking timely care reflects responsible health behavior rather than personal failure.
Finally, there is a significant risk for infection transmission, particularly when individuals are unaware of their infectious status, do not complete treatment, or resume sexual activity prematurely. Because many Chlamydia infections remain asymptomatic, patients may unknowingly transmit the organism to sexual partners, thereby contributing to persistent community spread (Detels et al., 2011; Torrone et al., 2014; Workowski et al., 2021). Nursing interventions in this area should emphasize partner notification, abstinence during treatment, adherence to therapy, and consistent use of barrier protection. Education and counseling remain central to reducing transmission and supporting broader public health prevention efforts.
2.1 Causes
Chlamydia infection arises from exposure to Chlamydia trachomatis, a bacterium classified within the Chlamydiaceae family and historically grouped under the Chlamydophila genus. This microorganism possesses distinctive biological characteristics that contribute to its pathogenicity and persistence within the human host. It is a gram-negative organism with an obligate intracellular life cycle, meaning that it can replicate only within living eukaryotic cells. Unlike many other bacteria, C. trachomatis lacks the ability to generate sufficient energy independently, which necessitates reliance on host cellular machinery for survival and replication. Its developmental cycle alternates between the infectious elementary body and the metabolically active reticulate body, enabling both transmission and intracellular proliferation (Murray & McKay, 2021; O’Connell & Ferone, 2016).
Transmission of C. trachomatis occurs primarily through direct contact with infected mucosal surfaces. The most common route is sexual transmission, including vaginal, anal, and oral contact with an infected individual. Vertical transmission may also occur during childbirth, leading to neonatal conjunctivitis or pneumonia (Workowski et al., 2021; Schachter et al., 1986; Tipple et al., 1979). The high prevalence of asymptomatic infections contributes substantially to its continued spread, as infected individuals may unknowingly transmit the pathogen to others. This silent transmission dynamic remains a central factor in the persistent global burden of Chlamydia infection (Detels et al., 2011; Hocking et al., 2023).
A critical aspect of the pathogenic diversity of C. trachomatis lies in its classification into multiple serovars, which are differentiated on the basis of antigenic variation. A total of 18 serovars have been identified, each associated with specific clinical manifestations and epidemiological patterns (Murray & McKay, 2021; Morré et al., 2000). These serovars demonstrate tissue tropism, meaning they preferentially infect particular anatomical sites, thereby influencing disease presentation and severity. Serovars A, B, Ba, and C are primarily associated with trachoma, a chronic ocular infection that remains endemic in several regions of Africa and Asia. Repeated infection can result in conjunctival inflammation, scarring, eyelid deformity, and eventual blindness if untreated (Habtamu et al., 2025). The persistence of these serovars in resource-limited settings further highlights the role of environmental and socioeconomic factors in disease transmission and progression (Habtamu et al., 2025).
In contrast, serovars D through K are predominantly responsible for urogenital infections. These strains infect the epithelial lining of the genital tract, producing conditions such as urethritis in males and cervicitis in females. If left untreated, these infections may ascend to the upper reproductive tract, resulting in complications such as pelvic inflammatory disease, infertility, and ectopic pregnancy (Hocking et al., 2023; Workowski et al., 2021; Pelvic inflammatory disease, 2012). These serovars are the most frequently encountered in clinical practice and remain a major focus of screening and prevention programs.
Serovars L1, L2, and L3 are linked to lymphogranuloma venereum (LGV), a more invasive form of Chlamydia infection. LGV is typically characterized by an initial genital lesion followed by spread to regional lymph nodes, resulting in marked lymphadenopathy and, in some cases, severe proctocolitis (Mabey & Peeling, 2002; Murray & McKay, 2021). This form of infection is more prevalent in tropical and subtropical regions and is often associated with specific high-risk populations. Overall, the causes of Chlamydia infection are closely related to the unique microbiological properties of C. trachomatis, its routes of transmission, and the diversity of its serovars, each of which contributes to distinct clinical syndromes and public health challenges.
2.2 Risk Factors
Urogenital infection caused by Chlamydia trachomatis remains a major public health concern because of its high prevalence and continued transmission among sexually active populations. It is recognized as one of the most frequently reported bacterial sexually transmitted infections in the United States and remains among the most common sexually transmitted infections globally (Torrone et al., 2014; Hocking et al., 2023; Zhao et al., 2025). The widespread nature of this infection reflects a complex interaction of biological susceptibility, behavioral exposure, and gaps in preventive healthcare access. Epidemiological evidence further suggests that the burden of disease is not evenly distributed, with certain demographic groups demonstrating disproportionately higher rates of infection (Currenti et al., 2024; Zhao et al., 2025).
Gender-based differences play a substantial role in the epidemiology of urogenital Chlamydia infection. The reported rate among women in the United States is approximately twice that observed in men. This disparity is likely influenced by a combination of biological and healthcare-related factors, including increased susceptibility of the female genital tract and more frequent screening among women, which leads to higher detection rates. By contrast, many infections in men remain undiagnosed because of asymptomatic presentation and lower participation in routine screening services (Torrone et al., 2014; Hocking et al., 2023).
Age is another critical determinant of risk, with adolescents and young adults showing the highest rates of infection. Among females, prevalence is especially elevated in those aged 15 to 24 years, a population characterized by both biological vulnerability and behavioral risk factors. Cervical ectopy, which is more common in younger women, increases susceptibility to infection by facilitating bacterial access to epithelial cells. In males, the highest rates are often observed between 20 and 24 years of age, reflecting patterns of sexual behavior and partner exposure (Hocking et al., 2023; Torrone et al., 2014; Zhao et al., 2025).
Behavioral factors also contribute substantially to the risk profile of Chlamydia infection. These include having multiple sexual partners, inconsistent condom use, unprotected sexual activity, and early initiation of sexual intercourse. A lack of awareness regarding sexually transmitted infections may further increase vulnerability, particularly among younger individuals and populations with limited access to sexual health education (Crichton et al., 2015; Workowski et al., 2021). Individuals with a prior history of sexually transmitted infections are also at elevated risk of reinfection, especially when partner treatment and follow-up care are incomplete or delayed (Currenti et al., 2024; Hao et al., 2025).
Limited access to healthcare services is another important determinant of risk. Populations with reduced access to screening, diagnostic testing, treatment, and follow-up care are more likely to experience undetected and untreated infections, thereby sustaining transmission within the community. Social and structural factors, including socioeconomic disadvantage, healthcare inequity, and inadequate sexual health infrastructure, further amplify this burden (Crichton et al., 2015; Owusu-Edusei et al., 2013). Overall, the interaction of demographic, biological, behavioral, and healthcare-related factors underscores the complexity of Chlamydia risk and highlights the importance of targeted prevention, routine screening, and early intervention strategies.
2.3 Assessment
Infection with Chlamydia trachomatis is associated with a broad spectrum of clinical manifestations involving both urogenital and extragenital systems. The organism exhibits a marked affinity for columnar epithelial cells, explaining its predilection for mucosal surfaces of the genital tract, rectum, conjunctiva, and oropharynx. Consequently, it contributes to multiple disease processes, including cervicitis, pelvic inflammatory disease, urethritis, epididymitis, prostatitis, and lymphogranuloma venereum. Beyond the urogenital tract, C. trachomatis is also implicated in extragenital conditions such as conjunctivitis, perihepatitis, pharyngitis, reactive arthritis, and proctitis (O’Connell & Ferone, 2016; Murray & McKay, 2021; Hocking et al., 2023). This wide clinical spectrum necessitates comprehensive assessment strategies that account for both localized and systemic involvement.
A defining characteristic of Chlamydia infection is its frequently asymptomatic course. A substantial proportion of infected individuals remain unaware of their condition, thereby functioning as silent reservoirs for ongoing transmission. This asymptomatic nature complicates clinical detection and delays intervention, ultimately increasing the likelihood of complications (Detels et al., 2011; Hocking et al., 2023). When symptoms do occur, they are often mild, nonspecific, and dependent on the anatomical site of infection. Therefore, clinical assessment must integrate patient history, risk evaluation, and targeted diagnostic testing to ensure accurate identification of the disease (Workowski et al., 2021).
Cervicitis represents one of the most common manifestations in women, yet it frequently presents with minimal or subtle clinical findings. Approximately 70% of affected women are asymptomatic or exhibit only mild symptoms, such as abnormal vaginal discharge, intermenstrual or postcoital bleeding, lower abdominal discomfort, and dysuria (Hocking et al., 2023; Torrone et al., 2014). The classic presentation of mucopurulent cervicitis—characterized by purulent endocervical discharge and cervical friability—is observed in only a minority of cases. This variability underscores the importance of maintaining a high index of suspicion, particularly in sexually active women within high-risk age groups.
Pelvic inflammatory disease develops when infection ascends from the lower genital tract to involve the uterus, fallopian tubes, and surrounding pelvic structures. This progression represents a serious complication with significant long-term consequences. Patients typically present with lower abdominal or pelvic pain, which may occur alone or in combination with signs of cervicitis. Clinical severity can vary widely, ranging from mild discomfort to severe inflammatory disease with systemic features. Early recognition is critical, as delayed diagnosis increases the risk of infertility, ectopic pregnancy, and chronic pelvic pain (Pelvic inflammatory disease, 2012; Workowski et al., 2021).
Urethritis is more commonly identified in men and represents a frequent clinical presentation of Chlamydia infection in this population. Differentiating chlamydial urethritis from gonococcal urethritis based solely on clinical features is often difficult due to overlapping symptoms, making laboratory confirmation essential. Patients typically report dysuria accompanied by urethral discharge that may appear white, gray, or clear. Although symptoms are often mild, untreated urethritis may lead to complications and continued transmission (O’Connell & Ferone, 2016; Workowski et al., 2021).
Epididymitis is another important manifestation in male patients, resulting from the extension of infection to the epididymis. It is characterized by unilateral testicular pain, tenderness, swelling, and occasionally hydrocele formation. Systemic symptoms such as fever may indicate a more advanced inflammatory response. Accurate clinical assessment is essential to differentiate this condition from other causes of acute scrotal pain, such as testicular torsion, which requires urgent intervention (Workowski et al., 2021).
Proctitis occurs primarily in individuals engaging in receptive anal intercourse and reflects infection of the rectal mucosa. Clinical features include rectal pain, mucopurulent discharge, and rectal bleeding. These symptoms may mimic other gastrointestinal disorders, emphasizing the importance of obtaining a detailed sexual history during assessment. In some cases, proctitis may coexist with other sexually transmitted infections, further complicating diagnosis (Crichton et al., 2015; Workowski et al., 2021).
Lymphogranuloma venereum represents a more invasive form of C. trachomatis infection. The initial stage is often marked by a small, painless genital ulcer that may go unnoticed, followed by the development of significant regional lymphadenopathy, particularly in the inguinal region. In advanced cases, lymph nodes may become tender, enlarged, and suppurative. The progression of LGV reflects deeper tissue invasion and a more pronounced inflammatory response compared to noninvasive serovars (Mabey & Peeling, 2002; Murray & McKay, 2021).
Extragenital manifestations further broaden the clinical relevance of Chlamydia infection. Conjunctivitis may occur through direct inoculation, particularly in neonates during childbirth or through autoinoculation in adults (Schachter et al., 1986; Mordhorst & Dawson, 1971). Perihepatitis, also known as Fitz-Hugh–Curtis syndrome, presents with right upper quadrant abdominal pain associated with pelvic inflammatory disease (Ekabe et al., 2017). Reactive arthritis represents an immune-mediated complication characterized by joint inflammation, urethritis, and conjunctivitis (Kobayashi & Kida, 2005). Pharyngitis may occur following oral exposure, while proctitis reflects rectal involvement as previously described.
Comprehensive assessment of Chlamydia infection therefore requires a multidimensional approach that integrates clinical evaluation with appropriate diagnostic testing. Given the high prevalence of asymptomatic infection, routine screening in high-risk populations remains essential. Early identification and treatment not only improve individual patient outcomes but also play a critical role in reducing transmission and preventing long-term complications (Hocking et al., 2023; Workowski et al., 2021).
2.4 Evaluation
The evaluation of Chlamydia trachomatis infection requires a structured approach that integrates clinical judgment with laboratory-based confirmation. Although certain manifestations of Chlamydia infection may present with recognizable clinical features, most cases cannot be definitively diagnosed on clinical grounds alone. Trachoma represents a notable exception, as it can often be identified through characteristic ocular findings, including chronic conjunctival inflammation, scarring, and progressive structural changes of the eyelid and conjunctiva (Habtamu et al., 2025; Mordhorst & Dawson, 1971). In contrast, other forms of Chlamydia infection, particularly those involving the urogenital tract, are associated with clinical syndromes that overlap with other infectious conditions, thereby necessitating confirmatory diagnostic testing to ensure accuracy (Taylor-Robinson, 1997; Hocking et al., 2023; Workowski et al., 2021).
Laboratory evaluation plays a central role in the diagnosis of urogenital Chlamydia infections. The current gold
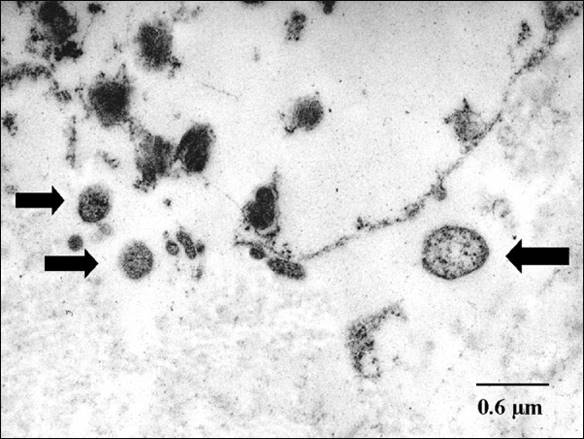
Figure 1. Transmission electron microscopy demonstrating intracellular chlamydial bodies within infected host cells.
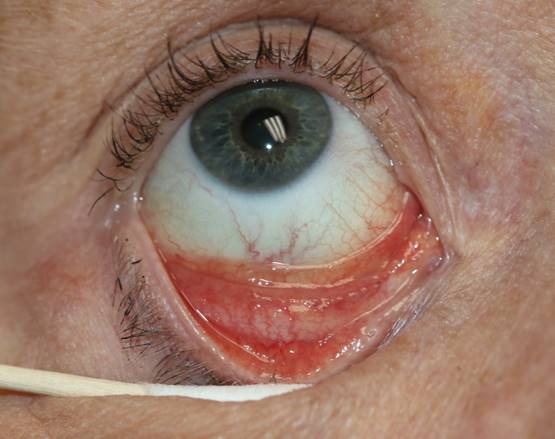
Figure 2. Follicular conjunctivitis associated with chlamydial infection. Inflammation may also be observed in viral infections and infectious mononucleosis.
standard is nucleic acid amplification testing (NAAT), which offers high sensitivity and specificity through the detection of C. trachomatis genetic material. This method has substantially improved diagnostic accuracy compared with earlier techniques and is widely recommended in current clinical practice (Taylor-Robinson, 1997; Hocking et al., 2023; Workowski et al., 2021). In women, NAAT is commonly performed using self-collected or clinician-obtained vaginal swabs, both of which have demonstrated strong diagnostic reliability. In men, first-catch urine samples are typically preferred, as they provide a noninvasive and effective means of identifying urethral infection. NAAT may also be applied to endocervical and urethral swabs when clinically indicated, particularly in symptomatic individuals or when more targeted sampling is required (Workowski et al., 2021). At the ultrastructural level, the intracellular nature of the organism can be appreciated through microscopy, where chlamydial bodies may be visualized within infected host cells (Figure 1).
Beyond urogenital specimens, NAAT can also be used for extragenital testing, including rectal and oropharyngeal samples, particularly in individuals with relevant exposure histories. This expanded diagnostic application improves the detection of infections that might otherwise remain unrecognized, thereby supporting more comprehensive and individualized patient management (Hocking et al., 2023; Workowski et al., 2021). The widespread adoption of NAAT reflects not only its superior diagnostic performance but also its adaptability across diverse clinical settings.
Alternative diagnostic methods are also available, although they are generally considered less sensitive or less practical than NAAT. Culture techniques, while historically regarded as definitive, are technically demanding and require specialized laboratory infrastructure, which limits their routine use. Rapid diagnostic tests offer the advantage of speed but often lack the sensitivity required for dependable screening and case identification. Serological testing may be useful in selected situations, such as retrospective exposure assessment or the evaluation of certain complications, but it is not routinely recommended for the diagnosis of acute genital infection. Antigen detection methods and genetic probe assays also exist; however, their performance varies, and they are generally not preferred when NAAT is available (Taylor-Robinson, 1997; Hocking et al., 2023).
In settings where diagnostic testing is unavailable or delayed, clinical evaluation becomes especially important. Under such circumstances, treatment may be initiated empirically on the basis of symptoms, risk profile, and the estimated likelihood of infection. This approach may be justified in high-risk populations or in situations where the consequences of untreated infection are potentially severe (Workowski et al., 2021). Nevertheless, reliance on clinical judgment alone carries the risk of both overtreatment and failure to identify alternative diagnoses, thereby reinforcing the need to expand access to accurate laboratory-based testing.
Effective evaluation of Chlamydia infection is essential not only for confirming diagnosis but also for guiding timely treatment, partner management, and prevention of onward transmission. Early and precise identification supports appropriate intervention, reduces the risk of reproductive and systemic complications, and strengthens broader public health efforts aimed at controlling the spread of this common infection (Hocking et al., 2023; Workowski et al., 2021; Hao et al., 2025). Similarly, ocular manifestations such as follicular conjunctivitis may provide clinically important visual clues in certain presentations of chlamydial disease (Figure 2)


