1. Introduction
The contemporary scientific landscape is being shaped, perhaps more strongly than ever, by two pressures that at first seem unrelated but are, in reality, deeply intertwined. One is the escalating demand for energy and materials in a world still heavily dependent on fossil resources. The other is the mounting public health burden imposed by antimicrobial resistance, a crisis that has begun to unsettle even the most routine assumptions about infection control and therapeutic reliability. The transition away from fossil-based industrial systems is no longer simply a climate objective; it is increasingly a biomedical and ecological necessity as well. Fossil energy continues to drive environmental degradation at a planetary scale, while renewable transitions remain uneven across sectors and regions (Kartal, 2022; Farghali et al., 2023). In that context, the bioeconomy has emerged not merely as a fashionable policy term, but as a broader rethinking of how biological resources can be used to replace extractive, linear production models with regenerative and circular ones (Priefer et al., 2017).
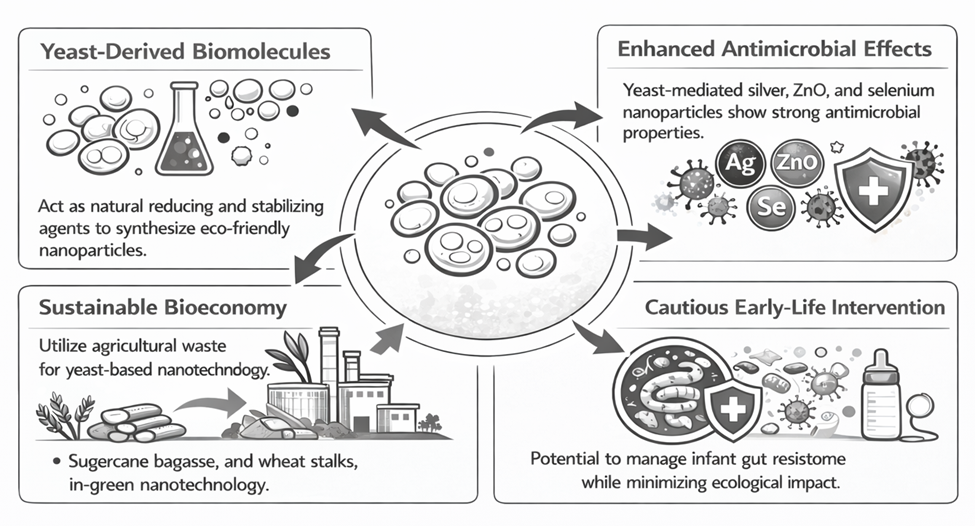
A circular bioeconomy depends, however, on a reliable and sustainable biomass base. That requirement sounds straightforward, yet it immediately raises questions of land use, feedstock competition, supply stability, and conversion efficiency. Biomass must not only be abundant; it must also be processable, affordable, and environmentally defensible. These concerns have made lignocellulosic residues particularly attractive, since they draw on agricultural and forestry by-products rather than primary food resources. Their appeal lies in both scale and versatility, offering a route to fuels, chemicals, and advanced materials while avoiding some of the ethical and economic tensions associated with first-generation biomass streams (Lewandowski, 2015; Ubando et al., 2020). Sugarcane bagasse, for example, has become a widely cited case of how a regional residue can serve both energy and non-energy applications within an integrated industrial system (Hofsetz & Silva, 2012).
Yet lignocellulosic biomass is, by nature, stubborn. Its resistance to deconstruction is not accidental; it is a structural consequence of the intimate association between cellulose, hemicellulose, and lignin in the plant cell wall. That architecture grants plants strength and resilience, but from a biorefinery perspective it creates a formidable barrier to hydrolysis, microbial conversion, and selective product recovery (Bajpai, 2016; Scheller & Ulvskov, 2010). Even when cellulose-rich fractions can be isolated, the route from raw biomass to functional product is rarely simple. Considerable effort must be invested in pretreatment before sugars, aromatics, or nanoscale materials can be efficiently generated. Recent work has therefore focused not only on biomass conversion as such, but on the pretreatment logic that determines whether downstream valorization will be economically and environmentally viable (Baruah et al., 2018; Haq et al., 2021).
This is precisely where the conversation broadens. Biomass is no longer being viewed solely as a substrate for fermentation or combustion; increasingly, it is being treated as a platform for advanced material design. Lignin, once handled largely as an inconvenient by-product, is now being reconsidered as a chemically rich feedstock for value-added products, including aromatic precursors, stabilizers, and nanoscale formulations (Ali et al., 2024; Lancefield et al., 2018). Likewise, cellulose-derived structures have drawn sustained interest because of their ability to yield nanocellulose and related materials with appealing mechanical, colloidal, and surface properties (George & Sabapathi, 2015; Trache et al., 2020). These developments suggest that biomass valorization is not confined to biofuel production; it also opens the door to functional materials relevant to packaging, catalysis, sensing, and antimicrobial technologies.
Nanotechnology, in turn, offers a compelling bridge between material innovation and biological application. Nanoparticles are attractive partly because scale changes behavior: at the nanoscale, materials often display altered reactivity, optical behavior, interfacial dynamics, and biological interactions. Still, the term “nanoparticle” itself carries environmental and toxicological implications, and those implications have prompted a broader discussion about how such materials should be defined, produced, and deployed responsibly (Auffan et al., 2009). Conventional synthesis routes often rely on aggressive chemicals, high-energy conditions, or reagents whose environmental cost undercuts the sustainability claims later attached to the final product. That tension has driven growing interest in green synthesis approaches, where biological systems or biomolecules act as reductants, stabilizers, templates, or capping agents.
Among the biological platforms explored for this purpose, yeasts occupy a particularly interesting position. They are metabolically versatile, relatively easy to cultivate, and already familiar to industrial biotechnology. More importantly, they produce an impressive range of extracellular and cell-associated biomolecules—proteins, polysaccharides, glycolipids, enzymes, pigments, and organic acids—that can mediate nanoparticle nucleation and stabilization under relatively mild conditions. The result is not simply a “greener” synthesis route in the narrow sense of avoiding harsh reagents. It may also yield nanoparticles with biologically meaningful surface features that influence dispersion stability, redox behavior, antimicrobial performance, and compatibility with living systems. Experimental studies have already shown that yeast-associated systems can support the synthesis of silver, zinc oxide, and selenium nanoparticles with noteworthy biological activities (Moghaddam et al., 2017; Salem, 2022).
The relevance of this field becomes even clearer when green nanotechnology is viewed through a biomedical lens. Antimicrobial resistance is not only a hospital problem or a late-stage infection problem; it is also a matter of ecological selection within the human microbiome, including very early in life. The infant gut microbiome develops during a period of extraordinary physiological vulnerability, and the accumulation of antimicrobial resistance determinants during that window may carry consequences that are both immediate and long-lasting. That makes the infant gut resistome an especially sensitive site of intervention. The challenge, though, is not merely to kill pathogens. It is to do so selectively, gently, and with enough biological sophistication that beneficial colonization and host-microbe signaling are not unnecessarily disrupted.
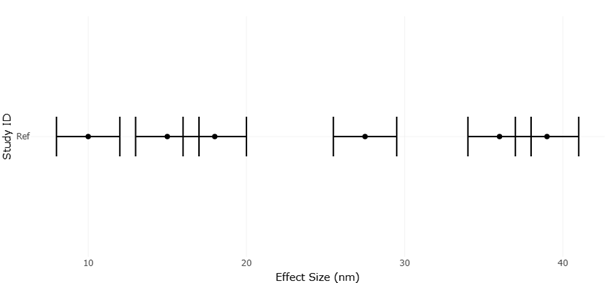
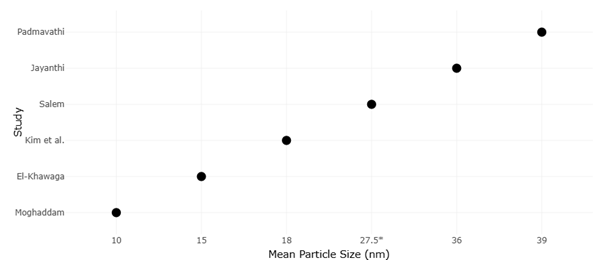
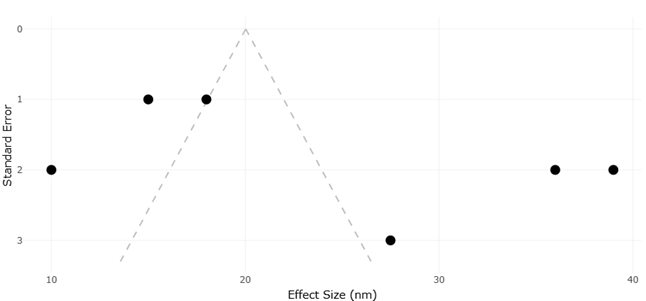
Here, yeast-derived nanomaterials begin to look less like a niche materials science topic and more like a candidate strategy for precision antimicrobial modulation. Studies using Saccharomyces cerevisiae and other yeast systems suggest that biosynthesized metal and metal oxide nanoparticles can exert meaningful antibacterial effects while being generated through routes that are, at least in principle, more biocompatible and less chemically burdensome than standard reduction-based methods (El-Khawaga et al., 2025; Kim et al., 2024). Baker’s yeast extract has also been used to fabricate selenium nanoparticles with antimicrobial efficacy, reinforcing the idea that yeast is not confined to one material class or one synthesis mode (Salem, 2022). Importantly, comparative work indicates that biologically and chemically synthesized nanoparticles may differ not only in particle size or morphology, but also in optical, structural, and antibacterial behavior, suggesting that synthesis route is itself a determinant of function rather than a merely procedural detail (Jayanthi et al., 2024; Padmavathi et al., 2022).
The sustainability argument remains equally important. Pretreatment and hydrolysis processes that liberate useful biomass fractions can also generate inhibitory compounds, including furans and phenolics, which complicate microbial conversion and downstream integration. This has encouraged the development of alternative pretreatment strategies designed to improve fractionation while limiting inhibitor formation or improving process compatibility. Approaches involving supercritical fluids, optimized acid or alkaline methods, and other intensified pretreatment systems are increasingly being explored for this reason (Escobar et al., 2020; Lorenci Woiciechowski et al., 2020). At the microbial level, S. cerevisiae has also been studied for its ability to tolerate and convert inhibitory compounds derived from lignocellulosic hydrolysates, a trait that becomes particularly relevant if biomass valorization and yeast-mediated nanomaterial production are to be linked within the same integrated framework (Almeida et al., 2007). Classical hydrolysis models and inhibitor studies still matter here, because they help explain why feedstock chemistry continues to shape the feasibility of green bioprocesses even when the final target is a nanomaterial rather than a fuel (Jacobsen & Wyman, 2000; Palmqvist & Hahn-Hägerdal, 2000).
What emerges from this convergence is a conceptually rich, if still developing, research space. On one side lies the logic of biomass conversion and circular material economies. On the other lies the urgent need for antimicrobial tools that are more selective, more adaptable, and perhaps less ecologically disruptive than conventional broad-spectrum approaches. Yeast-derived biomolecules sit at that interface. They connect renewable feedstocks to functional nanomaterials, and functional nanomaterials to potential biomedical applications. Recent discussions of lignocellulosic valorization in Europe and elsewhere only reinforce the timeliness of this connection, as bio-based production systems are increasingly expected to deliver not just fuels, but high-value products with measurable societal benefit (Güleç et al., 2024; Kulolo et al., 2025).
This narrative review was therefore developed to examine, in an integrated way, how yeast-derived biomolecules are being used in green nanoparticle synthesis, how those efforts relate to biomass valorization and sustainable processing, and why such materials may be relevant to the mitigation of the infant gut resistome. The aim is not to overstate translational readiness. Rather, it is to bring together scattered lines of evidence—industrial, biochemical, and antimicrobial—and ask whether they point toward a credible new framework for sustainable early-life microbiome intervention.