1. Introduction
Few enteric pathogens have reshaped modern hospital epidemiology as profoundly as Clostridioides difficile. Despite decades of clinical familiarity, this anaerobic, spore-forming bacterium continues to challenge clinicians and researchers alike. Once considered primarily an opportunistic pathogen associated with antibiotic exposure, C. difficile has gradually emerged as a complex ecological and clinical problem—one that sits at the intersection of antimicrobial therapy, microbial ecology, and host immunity. The organism was first described nearly a century ago when Hall and O’Toole identified a previously unrecognized anaerobe in the intestinal flora of newborn infants, naming it Bacillus difficilis because of the difficulty encountered in culturing it (Hall & O’Toole, 1935). In retrospect, that early observation hinted at a broader biological reality: this organism is remarkably resilient, capable of persisting in both environmental reservoirs and the human gut through the formation of highly resistant spores.
Over the past two decades, the epidemiology of C. difficile infection (CDI) has shifted in ways that are difficult to ignore. What was once regarded largely as a complication of antibiotic therapy is now recognized as a global healthcare threat. Incidence rates have risen across multiple regions, accompanied by increasing disease severity, higher recurrence rates, and mounting healthcare costs. These trends are not solely attributable to improved diagnostics; rather, they appear to reflect a genuine expansion in disease burden linked to demographic changes, widespread antibiotic use, and evolving bacterial virulence. Recent clinical analyses emphasize that CDI now represents one of the most common healthcare-associated infections worldwide, affecting diverse patient populations beyond traditional hospital settings (Markantonis et al., 2024; Vitiello et al., 2024). The persistence of spores in healthcare environments further complicates infection control, as they can withstand routine sanitation procedures and remain viable on surfaces for extended periods (McSharry et al., 2021).
The biological success of C. difficile lies partly in its unusual life cycle. Transmission typically occurs through ingestion of dormant spores that survive gastric acidity and reach the colon intact. Under normal circumstances, the indigenous gut microbiota prevents colonization through mechanisms collectively known as colonization resistance. However, antibiotic exposure—or, more broadly, any disruption of microbial community structure—can destabilize this protective barrier. In such altered conditions, C. difficile spores germinate and proliferate, giving rise to toxin-producing vegetative cells. The resulting toxins, primarily TcdA and TcdB, disrupt epithelial cytoskeletal integrity and trigger intense inflammatory responses within the colonic mucosa. This cascade of cellular injury and immune activation underlies the clinical manifestations of CDI, which range from mild diarrhea to fulminant colitis and toxic megacolon (Johnson et al., 2021; Lee, Yoo, & Yoon, 2024).
Yet the pathogenesis of CDI is not defined solely by these canonical toxins. Increasing attention has been directed toward additional virulence factors, particularly the binary toxin CDT. Although its precise contribution to disease progression remains debated, accumulating evidence suggests that CDT enhances bacterial adherence and modulates host immune responses in ways that amplify pathogenicity. Experimental studies have shown that the toxin suppresses protective eosinophilic responses within the intestinal mucosa, thereby facilitating bacterial persistence and tissue injury (Cowardin et al., 2016). Structural and biochemical analyses further indicate that targeting this toxin may hold therapeutic promise, especially in infections caused by hypervirulent strains (Abeyawardhane et al., 2021). These findings underscore a broader realization: CDI pathogenesis involves a multilayered interplay between microbial virulence mechanisms and host defense pathways.
The clinical burden of CDI is magnified by its tendency to recur. Even after successful treatment of an initial episode, approximately one quarter of patients experience relapse, and the likelihood of recurrence increases dramatically after subsequent infections. Recurrent CDI (rCDI) represents one of the most frustrating aspects of disease management, both for patients and healthcare providers. Conventional antibiotic therapy, while often effective in controlling acute infection, paradoxically perpetuates the underlying ecological disturbance within the gut microbiome. As a result, each therapeutic cycle may inadvertently create conditions conducive to further relapse (Louie et al., 2011; Johnson et al., 2021). Fidaxomicin, a narrow-spectrum macrocyclic antibiotic, partially addresses this limitation by reducing collateral damage to commensal microbes and inhibiting spore production, yet recurrence remains a significant concern (Babakhani et al., 2012).
These therapeutic limitations have prompted a gradual but profound shift in how CDI is conceptualized. Increasingly, researchers view the disease not merely as an infection caused by a single pathogen but as a manifestation of ecological imbalance within the intestinal microbiome. From this perspective, restoring microbial diversity becomes as important as suppressing bacterial growth. One of the most striking demonstrations of this principle is fecal microbiota transplantation (FMT), a procedure in which stool from healthy donors is introduced into the gastrointestinal tract of affected patients. Clinical trials have consistently reported remarkable efficacy, particularly in individuals with recurrent disease. In one randomized study, FMT significantly outperformed antibiotic therapy in achieving sustained remission (Hvas et al., 2019). Mechanistic investigations suggest that the procedure restores key metabolic functions of the gut microbiota, including bile acid transformations that inhibit C. difficile spore germination (Allegretti et al., 2016).
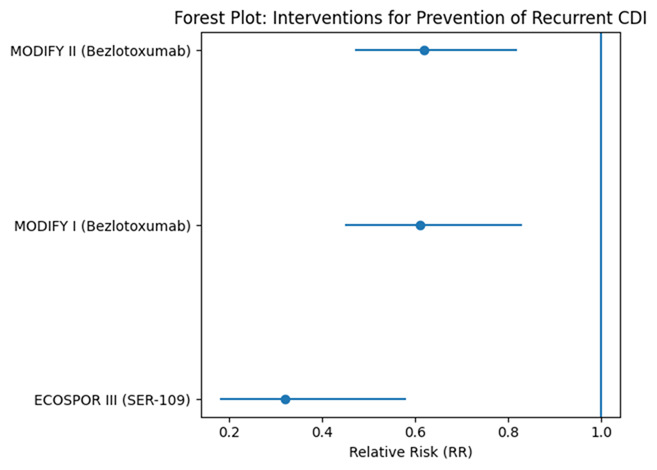
While FMT has proven transformative, it is not without challenges. Concerns regarding donor screening, pathogen transmission, and regulatory oversight have motivated the development of more standardized microbiome-based therapies. In recent years, live biotherapeutic products (LBPs) have emerged as a promising alternative. These formulations typically consist of carefully selected bacterial spores or microbial communities designed to reestablish ecological balance in the intestine. Clinical trials evaluating SER-109, an orally administered spore-based therapeutic, have demonstrated significant reductions in recurrence rates among patients with rCDI (Feuerstadt et al., 2022). Complementary studies further highlight the role of microbiome-restoring therapies in patients with complex comorbidities, emphasizing their potential relevance in real-world clinical populations (Berenson et al., 2023). Another example, REBYOTA™, has shown favorable safety and efficacy profiles across multiple prospective trials (Lee et al., 2023).
Beyond microbiome restoration, several additional therapeutic strategies are under investigation. Monoclonal antibodies targeting toxin B, such as bezlotoxumab, have been shown to reduce recurrence when administered alongside standard antibiotic therapy (Wilcox et al., 2017). Emerging approaches in nanomedicine also hold promise, particularly for targeting both vegetative cells and resilient spores. Nanoparticle-based formulations and synthetic polymers have demonstrated the capacity to inhibit spore germination or suppress bacterial growth in experimental systems (Chen et al., 2019; Liu et al., 2014). Similarly, engineered nanomaterials capable of disrupting spore germination pathways have been explored as potential adjunctive therapies (Lee et al., 2017). Although many of these technologies remain in early stages of development, they illustrate the expanding therapeutic landscape for CDI.
Underlying all these advances is a deeper appreciation of the host–microbiome–pathogen triad. The immune system does not merely respond to C. difficile; it interacts dynamically with the microbial ecosystem of the gut. Recent studies highlight the importance of innate lymphoid cells and microbial metabolites in maintaining intestinal homeostasis and limiting pathogen expansion (Abt et al., 2015; Gurung et al., 2024). When dysbiosis disrupts these regulatory networks, susceptibility to infection increases and recovery becomes more difficult. Consequently, therapies that restore microbial and immunological balance may offer the most sustainable solutions for long-term disease control.
Taken together, these developments suggest that CDI should no longer be viewed simply as an antibiotic-associated infection. Rather, it represents a multifaceted disorder shaped by microbial ecology, host immunity, and evolving bacterial virulence. As the global burden of disease continues to rise, innovative therapeutic strategies—particularly those targeting the microbiome—are becoming central to clinical management. This review examines the emerging challenges posed by Clostridioides difficile infection and explores the rapidly evolving landscape of microbiome-targeted therapeutics aimed at disrupting the cycle of recurrence and restoring intestinal ecological stability.